| ųą╬─├¹ĘQ | APP/ABPPĄĒĘ█śėļ─Ū░¾wĄ░░ū┐╣¾w |
| äe ├¹ | Soluble APP-alpha; Soluble APP-beta; A4 amyloid protein; A4; AAA; ABETA; ABPP; AD 1; AD1; Alzheimer disease 1; Alzheimer disease; Alzheimer's disease amyloid protein; Amyloid beta (A4) precursor protein; Amyloid beta A4 protein; Amyloid beta A4 protein precursor isoform b; Amyloid beta A4 protein precursor isoform c; Amyloid beta A4 protein precursor isoform a; Amyloid beta A4 protein precursor isoform b; Amyloid beta A4 protein precursor isoform c; Amyloid beta protein; Amyloid beta-peptide; Amyloid of aging and alzheimer disease; APP; APP I; APPI; Beta amyloid peptide; Cerebral vascular amyloid peptide; CTFgamma; CVAP; Human mRNA for amyloid A4 precursor of Alzheimer's disease; PN 2; PN II; PN2; PreA4; Protease nexin II; A4_HUMAN. |
蹊┐ŅIė“ą─č¬╣▄ ╝Ü░¹╔·╬’ ╔±Įø╔·╬’īW ą┼╠¢▐Dī¦ ╝Ü░¹Ą“═÷ Alzheimer's
┐╣¾wüĒį┤Rabbit
┐╦┬ĪŅÉą═Polyclonal
Į╗▓µĘ┤æ¬Human, Mouse, Rat, (predicted: Chicken, Dog, Pig, Cow, Horse, Rabbit, Sheep, )
«aŲĘæ¬ė├WB=1:500-2000 ELISA=1:500-1000 IHC-P=1:100-500 IHC-F=1:100-500 ICC=1:100 IF=1:100-500 Ż©╩»Ž×ŪąŲ¼ąĶū÷┐╣įŁą▐Å═Ż®
not yet tested in other applications.
optimal dilutions/concentrations should be determined by the end user.
Ęų ūė ┴┐72-83kDa
╝Ü░¹Č©╬╗╝Ü░¹║╦ ╝Ü░¹Ø{ ╝Ü░¹─ż ╝Ü░¹═Ō╗∙┘|
ąį ĀŅLiquid
ØŌ Č╚1mg/ml
├Ō ę▀ įŁKLH conjugated synthetic peptide derived from of human APP:381-480/770
üå ą═IgG
╝ā╗»ĘĮĘ©affinity purified by Protein A
ā” ┤µ ę║0.01M TBS(pH7.4) with 1% BSA, 0.03% Proclin300 and 50% Glycerol.
▒Ż┤µŚl╝■Shipped at 4Īµ. Store at -20 °C for one year. Avoid repeated freeze/thaw cycles.
PubMedPubMed
«aŲĘĮķĮBThis gene encodes a cell surface receptor and transmembrane precursor protein that is cleaved by secretases to form a number of peptides. Some of these peptides are secreted and can bind to the acetyltransferase complex APBB1/TIP60 to promote transcriptional activation, while others form the protein basis of the amyloid plaques found in the brains of patients with Alzheimer disease. Mutations in this gene have been implicated in autosomal dominant Alzheimer disease and cerebroarterial amyloidosis (cerebral amyloid angiopathy). Multiple transcript variants encoding several different isoforms have been found for this gene. [provided by RefSeq, Jul 2008].
Function:
Functions as a cell surface receptor and performs physiological functions on the surface of neurons relevant to neurite growth, neuronal adhesion and axonogenesis. Involved in cell mobility and transcription regulation through protein-protein interactions. Can promote transcription activation through binding to APBB1-KAT5 and inhibits Notch signaling through interaction with Numb. Couples to apoptosis-inducing pathways such as those mediated by G(O) and JIP. Inhibits G(o) alpha ATPase activity. Acts as a kinesin I membrane receptor, mediating the axonal transport of beta-secretase and presenilin 1. Involved in copper homeostasis/oxidative stress through copper ion reduction. In vitro, copper-metallated APP induces neuronal death directly or is potentiated through Cu(2+)-mediated low-density lipoprotein oxidation. Can regulate neurite outgrowth through binding to components of the extracellular matrix such as heparin and collagen I and IV. The splice isoforms that contain the BPTI domain possess protease inhibitor activity. Induces a AGER-dependent pathway that involves activation of p38 MAPK, resulting in internalization of amyloid-beta peptide and leading to mitochondrial dysfunction in cultured cortical neurons.
Beta-amyloid peptides are lipophilic metal chelators with metal-reducing activity. Bind transient metals such as copper, zinc and iron. In vitro, can reduce Cu(2+) and Fe(3+) to Cu(+) and Fe(2+), respectively. Beta-amyloid 42 is a more effective reductant than beta-amyloid 40. Beta-amyloid peptides bind to lipoproteins and apolipoproteins E and J in the CSF and to HDL particles in plasma, inhibiting metal-catalyzed oxidation of lipoproteins. Beta-APP42 may activate mononuclear phagocytes in the brain and elicit inflammatory responses. Promotes both tau aggregation and TPK II-mediated phosphorylation. Interaction with overexpressed HADH2 leads to oxidative stress and neurotoxicity.
Appicans elicit adhesion of neural cells to the extracellular matrix and may regulate neurite outgrowth in the brai.
The gamma-CTF peptides as well as the caspase-cleaved peptides, including C31, are potent enhancers of neuronal apoptosis.
N-APP binds TNFRSF21 triggering caspase activation and degeneration of both neuronal cell bodies (via caspase-3) and axons (via caspase-6).
Subunit:
Binds, via its C-terminus, to the PID domain of several cytoplasmic proteins, including APBB family members, the APBA family, MAPK8IP1, SHC1 and, NUMB and DAB. Binding to DAB1 inhibits its serine phosphorylation (By similarity). Also interacts with GPCR-like protein BPP, FPRL1, APPBP1, IB1, KNS2 (via its TPR domains), APPBP2 (via BaSS) and DDB1. In vitro, it binds MAPT via the MT-binding domains. Associates with microtubules in the presence of ATP and in a kinesin-dependent manner. Interacts, through a C-terminal domain, with GNAO1. Amyloid beta-42 binds CHRNA7 in hippocampal neurons. Beta-amyloid associates with HADH2. Soluble APP binds, via its N-terminal head, to FBLN1. Interacts with CPEB1 and AGER. Interacts with ANKS1B and TNFRSF21. Interacts with ITM2B. Interacts with ITM2C. Interacts with IDE. Can form homodimers; this is promoted by heparin binding.
Subcellular Location:
Membrane; Single-pass type I membrane protein. Membrane, clathrin-coated pit. Note=Cell surface protein that rapidly becomes internalized via clathrin-coated pits. During maturation, the immature APP (N-glycosylated in the endoplasmic reticulum) moves to the Golgi complex where complete maturation occurs (O-glycosylated and sulfated). After alpha-secretase cleavage, soluble APP is released into the extracellular space and the C-terminal is internalized to endosomes and lysosomes. Some APP accumulates in secretory transport vesicles leaving the late Golgi compartment and returns to the cell surface. Gamma-CTF(59) peptide is located to both the cytoplasm and nuclei of neurons. It can be translocated to the nucleus through association with APBB1 (Fe65). Beta-APP42 associates with FRPL1 at the cell surface and the complex is then rapidly internalized. APP sorts to the basolateral surface in epithelial cells. During neuronal differentiation, the Thr-743 phosphorylated form is located mainly in growth cones, moderately in neurites and sparingly in the cell body. Casein kinase phosphorylation can occur either at the cell surface or within a post-Golgi compartment.
Tissue Specificity:
Expressed in all fetal tissues examined with highest levels in brain, kidney, heart and spleen. Weak expression in liver. In adult brain, highest expression found in the frontal lobe of the cortex and in the anterior perisylvian cortex-opercular gyri. Moderate expression in the cerebellar cortex, the posterior perisylvian cortex-opercular gyri and the temporal associated cortex. Weak expression found in the striate, extra-striate and motor cortices. Expressed in cerebrospinal fluid, and plasma. Isoform APP695 is the predominant form in neuronal tissue, isoform APP751 and isoform APP770 are widely expressed in non-neuronal cells. Isoform APP751 is the most abundant form in T-lymphocytes. Appican is expressed in astrocytes.
Post-translational modifications:
Proteolytically processed under normal cellular conditions. Cleavage either by alpha-secretase, beta-secretase or theta-secretase leads to generation and extracellular release of soluble APP peptides, S-APP-alpha and S-APP-beta, and the retention of corresponding membrane-anchored C-terminal fragments, C80, C83 and C99. Subsequent processing of C80 and C83 by gamma-secretase yields P3 peptides. This is the major secretory pathway and is non-amyloidogenic. Alternatively, presenilin/nicastrin-mediated gamma-secretase processing of C99 releases the amyloid beta proteins, amyloid-beta 40 (Abeta40) and amyloid-beta 42 (Abeta42), major components of amyloid plaques, and the cytotoxic C-terminal fragments, gamma-CTF(50), gamma-CTF(57) and gamma-CTF(59).
Proteolytically cleaved by caspases during neuronal apoptosis. Cleavage at Asp-739 by either caspase-6, -8 or -9 results in the production of the neurotoxic C31 peptide and the increased production of beta-amyloid peptides.
N- and O-glycosylated. O-linkage of chondroitin sulfate to the L-APP isoforms produces the APP proteoglycan core proteins, the appicans. The chondroitin sulfate chain of appicans contains 4-O-sulfated galactose in the linkage region and chondroitin sulfate E in the repeated disaccharide region.
Phosphorylation in the C-terminal on tyrosine, threonine and serine residues is neuron-specific. Phosphorylation can affect APP processing, neuronal differentiation and interaction with other proteins. Phosphorylated on Thr-743 in neuronal cells by Cdc5 kinase and Mapk10, in dividing cells by Cdc2 kinase in a cell-cycle dependent manner with maximal levels at the G2/M phase and, in vitro, by GSK-3-beta. The Thr-743 phosphorylated form causes a conformational change which reduces binding of Fe65 family members. Phosphorylation on Tyr-757 is required for SHC binding. Phosphorylated in the extracellular domain by casein kinases on both soluble and membrane-bound APP. This phosphorylation is inhibited by heparin.
Extracellular binding and reduction of copper, results in a corresponding oxidation of Cys-144 and Cys-158, and the formation of a disulfide bond. In vitro, the APP-Cu(+) complex in the presence of hydrogen peroxide results in an increased production of beta-amyloid-containing peptides.
Trophic-factor deprivation triggers the cleavage of surface APP by beta-secretase to release sAPP-beta which is further cleaved to release an N-terminal fragment of APP (N-APP).
Beta-amyloid peptides are degraded by IDE.
DISEASE:
Defects in APP are the cause of Alzheimer disease type 1 (AD1) [MIM:104300]. AD1 is a familial early-onset form of Alzheimer disease. It can be associated with cerebral amyloid angiopathy. Alzheimer disease is a neurodegenerative disorder characterized by progressive dementia, loss of cognitive abilities, and deposition of fibrillar amyloid proteins as intraneuronal neurofibrillary tangles, extracellular amyloid plaques and vascular amyloid deposits. The major constituent of these plaques is the neurotoxic amyloid-beta-APP 40-42 peptide (s), derived proteolytically from the transmembrane precursor protein APP by sequential secretase processing. The cytotoxic C-terminal fragments (CTFs) and the caspase-cleaved products such as C31 derived from APP, are also implicated in neuronal death.
Defects in APP are the cause of amyloidosis cerebroarterial Dutch type (AMYLCAD) [MIM:605714]; also known as hereditary cerebral hemorrhage with amyloidosis Dutch type (HCHWAD). AMYLCAD is a hereditary localized amyloidosis due to amyloid-beta A4 peptide(s) deposition in the cerebral vessels. Beta-APP40 is the predominant form of cerebrovascular amyloid. Amyloid is not found outside the nervous system. The principal clinical characteristics are recurrent cerebral and cerebellar hemorrhages, recurrent strokes, cerebral ischemia, cerebral infarction, and progressive mental deterioration. Onset of the disease is in middle age (44 to 60 years). Patients develop cerebral hemorrhage because of the severe cerebral amyloid angiopathy. Parenchymal amyloid deposits are rare and largely in the form of pre-amyloid lesions or diffuse plaque-like structures. They are Congo red negative and lack the dense amyloid cores commonly present in Alzheimer disease.
Defects in APP are the cause of amyloidosis cerebroarterial Italian type (AMYLCAIT) [MIM:605714]. AMYLCAIT is a hereditary localized amyloidosis due to amyloid-beta A4 peptide(s) deposition in the cerebral vessels, resulting in cerebral amyloid angiopathy. Amyloid is not found outside the nervous system. It is a condition very similar to AMYLCAD, but the clinical course is less severe. Patients manifest mild cognitive decline, recurrent strokes, and epilepsy in some cases. There are extensive amyloid deposits in leptomeningeal and cortical vessels and, to a lesser extent, in the neuropil of the cerebral cortex, in the absence of neurofibrillary tangles.
Defects in APP are the cause of amyloidosis cerebroarterial Iowa type (AMYLCAIW) [MIM:605714]. AMYLCAIW is a hereditary amyloidosis due to amyloid-beta A4 peptide(s) deposition. Patients have progressive aphasic dementia, leukoencephalopathy, and occipital calcifications.
Similarity:
Belongs to the APP family.
Contains 1 BPTI/Kunitz inhibitor domain.
SWISS:
P05067
Gene ID:
351
Database links:
Entrez Gene: 351 Human
Entrez Gene: 11820 Mouse
Entrez Gene: 54226 Rat
Omim: 104760 Human
SwissProt: P05067 Human
SwissProt: P12023 Mouse
SwissProt: P08592 Rat
Unigene: 434980 Human
Unigene: 277585 Mouse
Unigene: 489029 Mouse
Unigene: 490986 Mouse
Unigene: 2104 Rat
Important Note:
This product as supplied is intended for research use only, not for use in human, therapeutic or diagnostic applications.
APPŻ©Amyloid protein precursorŻ®ĄĒĘ█śėĄ░░ūŪ░¾wĄ░░ū╩Ūę╗ĘN┤¾Ą─╝Ü░¹─żĄ░░ūŻ¼ŲõNČ╦╔ņ│÷ė┌╝Ü░¹═ŌĪŻįōĄ░░ūįŌĄĮŲŲē─▓óßīĘ┼│÷42éĆ░▒╗∙╦ߥ─β-ĄĒĘ█śėļ─Ż¼į┌└Ž─Ļ░▀║═č¬╣▄ųą▒╗░l¼FĪŻ▀@ą®«É│ŻĄ░░ū┐╔─▄ĘeŠ█Ż¼Č°ī¦ų┬╝Ü░¹╩¦š{║═╔±Įøį¬Ą─ōpé¹ĪŻ
į┌蹊┐APP┼cADĄ─ĻPŽĄųąŻ¼įō┐╣¾w╩«Ęųųžę¬ĪŻ
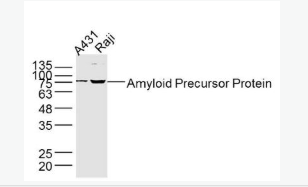
| «aŲĘłDŲ¼ | 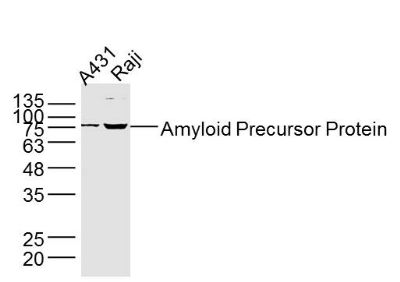 Sample: A431 Cell (Human) Lysate at 40 ug Raji Cell (Human) Lysate at 40 ug Primary: Anti- Amyloid Precursor Protein (bs-0112R) at 1/300 dilution Secondary: IRDye800CW Goat Anti-Rabbit IgG at 1/20000 dilution Predicted band size: 72-83 kD Observed band size: 75 kD 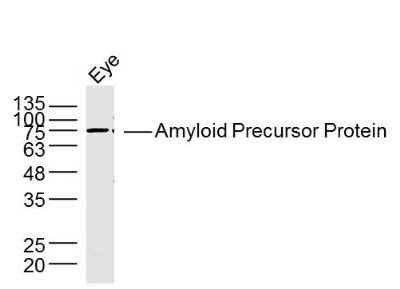 Sample: Eye (Rat) Lysate at 40 ug Primary: Anti- Amyloid Precursor Protein (bs-0112R) at 1/300 dilution Secondary: IRDye800CW Goat Anti-Rabbit IgG at 1/20000 dilution Predicted band size: 72-83 kD Observed band size: 75 kD 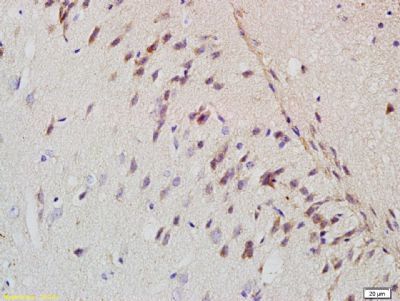 Tissue/cell: rat brain tissue; 4% Paraformaldehyde-fixed and paraffin-embedded; Antigen retrieval: citrate buffer ( 0.01M, pH 6.0 ), Boiling bathing for 15min; Block endogenous peroxidase by 3% Hydrogen peroxide for 30min; Blocking buffer (normal goat serum,C-0005) at 37Īµ for 20 min; Incubation: Anti-Amyloid Precursor Protein Polyclonal Antibody, Unconjugated(bs-0112R) 1:200, overnight at 4°C, followed by conjugation to the secondary antibody(SP-0023) and DAB(C-0010) staining 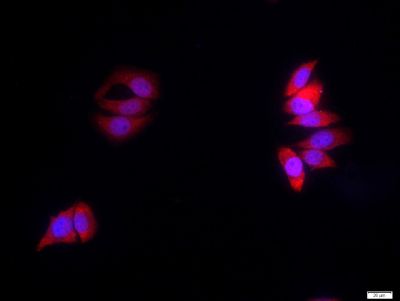 Tissue/cell: MCF7 cell; 4% Paraformaldehyde-fixed; Triton X-100 at room temperature for 20 min; Blocking buffer (normal goat serum, C-0005) at 37°C for 20 min; Antibody incubation with (Amyloid Precursor Protein) polyclonal Antibody, Unconjugated (bs-0112R) 1:100, 90 minutes at 37°C; followed by a FITC conjugated Goat Anti-Rabbit IgG antibody at 37°C for 90 minutes, DAPI (blue, C02-04002) was used to stain the cell nuclei. |